Growing Sweetpotatoes in Western Washington
Sweetpotato Pests and Diseases
Authors: Laura Schulz1, Laurel Moulton2, Samantha Grieger2, Jordan White1, Jessica Weaver1, Srijana Shrestha3, Carol Miles1
Affiliation: Washington State University, 1Northwestern Washington Research and Extension Center, Mount Vernon; 2Extension; 3Oregon State University Extension
https://vegetables.wsu.edu/sweetpotato/
January 2026
Introduction
Wireworms (Agriotes lineatus, A. obscurus, and Limonius canus) are the primary insect pest impacting sweetpotato root quality in western Washington. In 2025, 33% of farmers and gardeners trialing sweetpotatoes reported damage by wireworms, though only 6% reported moderate or severe damage. Wireworms are the immature (larval) stage of several native and introduced click beetles (family Elateridae) (Figure 1). Feeding holes are often superficial and can heal during the curing process but the cosmetic damage makes roots unmarketable (Figure 2). Additionally, dirt can get stuck in the holes created by wireworms, requiring additional scrubbing before cooking.


Field Selection to Avoid Wireworms
Wireworm populations generally increase in fields with continuous pasture or grass crops and decrease in fields with annual vegetable production (Poggi et al. 2021). To reduce the likelihood of wireworm damage, plant sweetpotatoes in a field that has been in annual vegetable production (Figure 4). Do not grow sweetpotatoes in a field that has recently had a grass or grain crop or that was recently planted in pasture. Avoid planting on field edges that are weedy or have grassy margins. Additionally, avoid fields that have a history of significant wireworm damage.
Other Insect Pests
Spotted Cucumber Beetle
Spotted cucumber beetle (Diabrotica undecimpunctata) larvae feed on sweetpotato roots, causing damage that is similar to wireworms while adult beetles cause superficial damage to leaves. Wireworm-resistant varieties are also resistant to spotted cucumber beetle larvae.
Foliar Insect Pests
Most sweetpotato pests of significance are larvae and grubs that attack roots. Sweetpotatoes can tolerate minor defoliation without yield loss, so caterpillars and chewing insects that feed on leaves and vines are rarely a problem. While most foliar pests are not cause for concern, aphids, whiteflies, and other sucking insects can be vectors for viruses.
Slugs
Slugs are considered a minor pest on sweetpotato and they will feed opportunistically on leaves and roots that protrude above the ground, with damage increasing towards the end of the season with fall rains and cooler weather. High humidity caused by irrigation, damp weather, and closed plant canopies favor slug activity. No-till or low-till practices, build-up of plant debris in the field, the use of mulches, and the use of wood borders to create raised beds also contribute to increasing slug populations by providing habitat and moist conditions.
Slug populations can be reduced by tillage and taking measures to reduce moist conditions. Harvesting sweetpotatoes early in the fall may prevent damage caused to roots that protrude from the ground later in the season. Small bait pellets that contain cereal bran or flour as attractants are the most effective. Baits must be applied according to the label instructions in locations where slugs will encounter them. Bread dough balls and cucumber slices have been shown to be effective attractants, but must be paired with physical removal of gathered slugs. See Pacific Northwest Insect Management Handbook for more detailed information.
Vertebrate Pests
Deer and Rabbits
Deer and rabbits are attracted to sweetpotato greens. In 2025, 20% of farmers and gardeners trialing sweetpotatoes reported damage by deer and 6% reported damage by rabbits. Without exclusion, they will graze plants down to the ground throughout the growing season, leading to crop failure. Cover plants with floating row cover or plastic slitted tunnels to exclude both deer and rabbits. If using fencing, it must be at least 8 feet high to exclude deer. For rabbits, a 2 to 3 foot fence made of 1-inch mesh with the bottom buried 6 inches deep is sufficient.
Voles, Rats, and Mice
Voles (Microtus spp.) and Norway rats (Ratus norvegicus) have been reported by farmers to be the most severe pests of sweetpotato crops in western Washington. In 2025, 37% of farmers and gardeners trialing sweetpotatoes reported issues with voles and 16% reported issues with rats. Voles and rats may consume whole or portions of sweetpotato roots (Figure 5). These rodents dig tunnels and build burrows under plastic mulch or other weed prevention soil covers where they are protected from predators and have easy access to the crop. It is important to differentiate between damage caused by voles versus rats because management tactics are different. Voles create runways along the top of sweetpotato beds under the plastic mulch, with their tunnels and burrows around sweetpotato roots. Norway rats also tunnel in the ground, but their burrows are lined with fibrous material such as leaves, shredded weed cloth, or plastic mulch (Figure 6).
Vole populations are irregularly cyclic depending on predator populations, weather, and quality of food and habitat. Minor population peaks occur about every 4 to 6 years and larger peaks occur every 10 to 12 years. Voles have the capacity to eat their body weight in food each day (Gunn et al. 2022). Rat populations are often more influenced by the availability of food and shelter and nearby human activity (Quinn 2024).
Reduce ground cover to deter rats and voles from entering a field. Remove vegetation taller than 6 inches, do not pile brush next to field edges, do not plant next to hedgerows, and mow and till field margins to minimize cover.
Encourage predators such as raptors, including hawks and owls, by providing raptor poles and other perching spots. Other natural enemies include garter snakes (for voles), coyotes, foxes, weasels and domestic cats. However, encouraging predators alone is not sufficient for managing voles or rats, since predators cannot keep up with the breeding rate of rodents (Pehling 2013).
Common mouse and rat traps (un-baited or baited with apple or peanut butter) are legal in Washington State. Use a mouse trap for trapping voles, while larger rat traps are needed for rats. Set traps for voles perpendicular inside vole runways, with at least one trap per 100 square feet. Keep all traps covered to protect birds and other off-target wildlife (Pehling 2013). Farmers in western Washington have reported that slightly earlier harvest dates can help minimize rodent damage to sweetpotatoes.
While there is limited research on their efficacy in sweetpotatoes, some farmers have experimented with blood meal-based animal repellants to deter voles and rats. Common granular formulations include 100% bloodmeal (which is also sold as a nitrogen fertilizer and typically contains between 13% and 14% nitrogen) or a wettable powder with 15% other “sticker” ingredients that makes the product stay in place for longer. To be most effective, the product needs to be placed at the crown of the plant and/or under the plastic mulch. Since sweetpotatoes respond to nitrogen fertilization by favoring leaf production over root production, care should be taken when experimenting with using bloodmeal as a rodent deterrent so as not to over fertilize the crop.
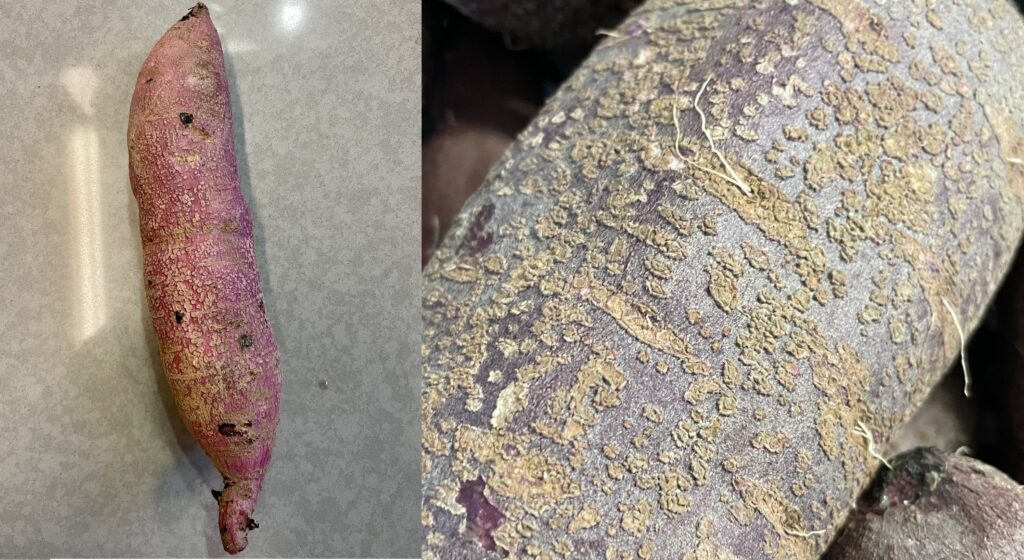
Prevention and sanitation are the best tools for avoiding crop infection. Scurf can be spread through the use of infected sweetpotato roots for slip production. When purchasing slips from a supplier, call and ask if they have had any scurf in their fields. Only order slips from companies that can guarantee disease-free plant material. Do not save roots that are visibly infected with scurf for slip production.
Infected roots should be cured and stored separately from ones that are not infected because the humid conditions needed for curing can cause the fungus to produce spores and spread. Crates and other farm tools that come into contact with scurf-infested roots should be sanitized. Sanitize curing chambers and storage areas to help keep inoculum to a minimum. Rotate sweetpotatoes out of infected fields for 2 to 4 years. The fungus persists longer in soils that are higher in organic matter. Field bindweed (Convolvulus arvensis) and morning glory (Calystegia × lucana) are common weeds that are in the same plant family as sweetpotato, and are alternative hosts of scurf.
Soft Rot
Soft rot caused by Rhizopus stolonifer is the most common and significant disease of sweetpotatoes in storage. The pathogen enters sweetpotato tissue through any type of wound, but particularly through bruises from rough handling during harvest and transport. Moist and cold soil conditions at harvest can cause sweetpotatoes to be more vulnerable to infection.
Soft rot causes watery, soft decay with a distinct fermented sweet smell that may attract fruit flies. Rot advances rapidly and roots become completely inedible within a few days. Masses of gray to white fungal mycelium and black spore structures can be seen growing on the skin and out of any injuries on decaying roots.
Minimizing injuries and curing roots in a timely manner is essential to reducing post-harvest disease. Curing seals wounds, providing a barrier against fungus colonization. Large, ‘crushing’ wounds provide more surface area for infection than clean cuts caused by sharp tools. Maintaining proper temperature (55–60°F), humidity (80–90%) and ventilation in storage areas will limit disease development. Minimize disease inoculum by sanitizing packing, sorting and storage equipment that may have come in contact with infected roots, and using clean water if washing sweetpotatoes after harvest. Some sweetpotato varieties, like Beauregard, have high resistance to soft rot (Benedict et al. 2009). Consider harvesting 1–2 weeks or earlier to reduce issues with soft rot and other storage diseases.
Foliar Disease
Foliar diseases, such as Alternaria leaf spot, caused by Alternaria spp. of fungi, have been an occasional problem in greenhouse or high tunnel grown sweetpotatoes in western Washington. Foliar diseases are best managed by avoiding overhead irrigation, and adequately venting greenhouses and high tunnels to prevent humid conditions and condensation.
Viruses
More than 30 viruses are known to infect sweetpotatoes, though only a few are important economically. Sweetpotato Virus Disease, a co-infection by 2 viruses, including sweetpotato feathery mottle virus (SPFMV) and sweetpotato chlorotic stunt virus (SPCSV), is the most important disease of sweetpotatoes worldwide. SPFMV is not harmful on its own, but when combined with SPCSV to cause Sweetpotato Virus Disease, symptoms include stunting, dwarfing, chlorosis, discoloration, and puckering.
Viruses can be spread by insects such as aphids and white flies, and by contact between plants. Removal of infected plants can be an effective method to prevent virus spread, and plants with symptoms should be removed immediately from the growing area. After SPVD-infected plants are removed, remaining plants should be monitored closely for symptoms. Control of virus vectors such as aphids during slip production can help reduce virus spread.
Viruses, especially SPVD, may be latent in storage roots, and even asymptomatic plants infected with SPVD can still produce slips that have severe symptoms. When producing slips, sterilize cutting tools in between roots to reduce the spread of infection, even if plants are asymptomatic. Purchasing certified disease-free slips or propagating slips on-farm with disease-free roots can help minimize virus occurrence. New propagation stock should be purchased when loss of vigor occurs within the crop or viral symptoms appear.
To date, there is no evidence of significant presence of sweetpotato viruses in Washington State. It is essential that only certified virus-free plant material is brought into the state to avoid issues in the future.
Physiological Diseases
Corky Root
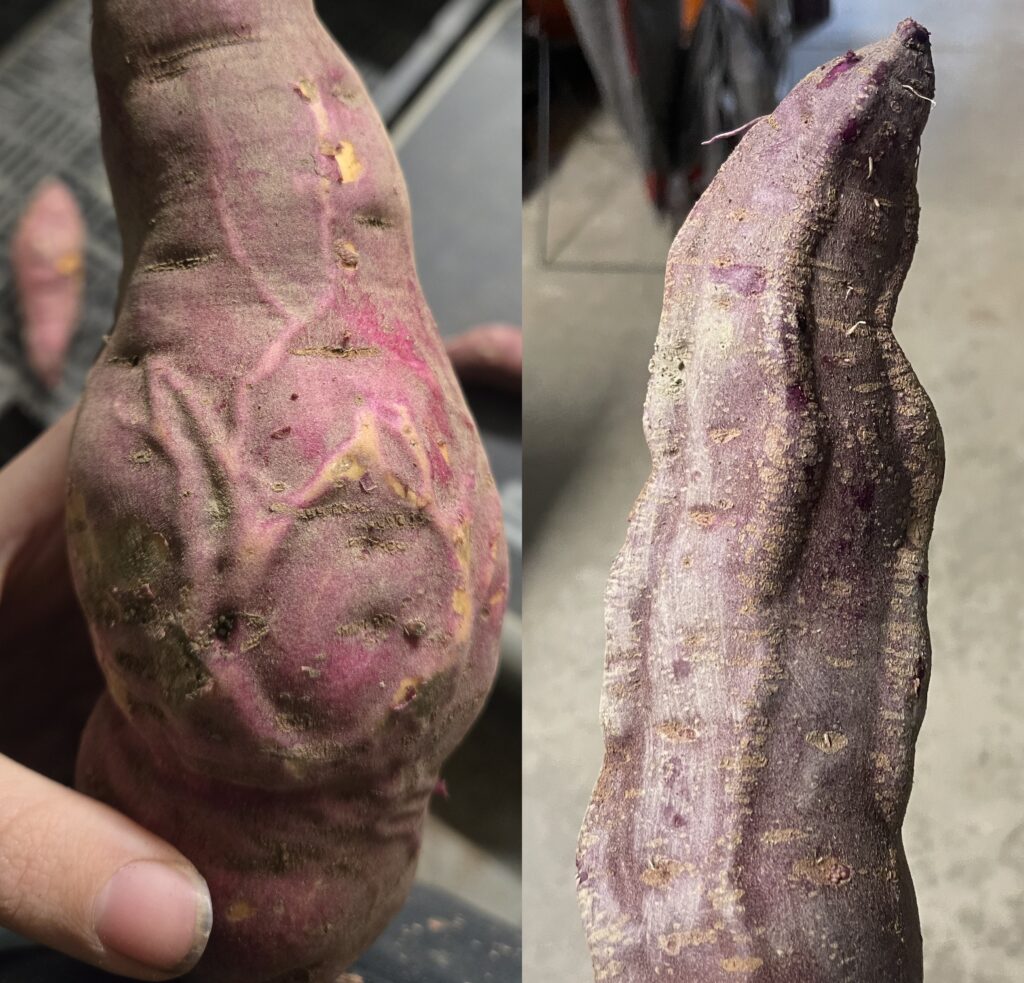
Corky root, also known as oedema or water blisters, occurs when lenticels (pores) in the skin of the root swell, resulting in small, raised corky lumps (Figure 9). Corky root is seen on roots grown in overly wet soil. Some sweetpotato varieties, like Monaco and Purple Majesty, are particularly susceptible to this disorder, while others, like Covington, are more resistant (North Carolina State University n.d.). Enlarged lenticels do not impact edible quality but may reduce market value if severe (Eckman et al. 2015). Earlier harvest to avoid on-set of fall rain and avoiding over-irrigation can help prevent corky root.
Water Edema
Overwatering can lead to several issues including water edema which is caused by the roots taking up more water than the leaves lose through transpiration. Symptoms include white crust or powder, indentions, or blisters as the excess water bursts the plant cells. There are two approaches to treating sweetpotatoes for water edema. First, increase leaf transpiration by lowering the humidity – this can be done by removing the clear plastic domes, increasing air circulation by spacing out trays or using a fan, or increasing the ambient temperature. Second, decrease the water intake by ensuring proper drainage in the plastic flats or reducing watering frequency.
Fasciation
Fasciated sweetpotatoes vines are flat, wide and strap-like, and may look like multiple vines fused together (Figure 12). Fasciation in sweetpotatoes is not “contagious” and does not spread through plant populations. Causes of fasciation can be hormonal imbalances, a genetic mutation, environmental factors, bacterial infection or physical damage to the growing point (Pscheidt and Putnam 2023). In sweetpotatoes, the specific cause is unknown and plant production appears to be unaffected (Eckman et al. 2015).

Root Splitting
Sweetpotatoes may split along the length of the root in response to water stress or other conditions. Growth cracks form when roots experience dry periods in the soil followed by heavy rain or irrigation events. Other causes of root splitting may be from low doses of herbicides or viral infections. Splits that have formed during early growth will heal over. Root splitting can be avoided by watering consistently and tapering off watering near the end of the season.
Cold Damage
Chilling injury occurs in sweetpotatoes exposed to temperatures below 50°F, but symptoms may not manifest for weeks or months after damage occurs. The most common symptoms of chilling injury are surface pitting (large round sunken patches), reduced storage life and increased susceptibility to rot. Chilling also accelerates dry matter loss, which may lead to internal tissue breakdown and can impact the taste, texture and color of the flesh.
The severity of injury depends on the temperature and the length of direct exposure. Unharvested sweetpotatoes will not be injured by low air temperatures, or even mild frost, as long as the soil temperature remains above damaging levels. Chilling injury is also cumulative, and severe damage may occur after repeated short exposures to damaging temperatures (Edmunds et al. 2008). For this reason, care must be taken to provide supplemental heat when curing sweetpotatoes in a high tunnel or greenhouse when night temperatures fall below 50°F.
References
Benedict LF, Villordon AO, Ferrin DM, Picha DH, Clark CA, Smith T, Labonte DR. 2009. Managing Rhizopus soft rot on sweet potatoes. Louisiana Agriculture Magazine, Spring 2009.
Eckman J, Lovatt J. 2015 Pests, diseases and disorders of sweetpotatoes: A field identification guide. Horticulture Innovation Australia Limited.
Edmunds B, Boyette M, Clark C, Ferrin D, Smith T, Holmes G. 2008. Postharvest handling of sweetpotatoes. AG-413-10-B.
Gunn D, Hirnyck R, Shewmaker G, Takatori S, Ellis LT. 2022. Meadow voles and pocket gophers: Management in lawns gardens and croplands (PNW0627). Pacific Northwest Extension Publications. https://www.uidaho.edu/-/media/uidaho-responsive/files/extension/publications/pnw/pnw0627.pdf?la=en&rev=e75b266efbcf44119f4698ad30373a75.
North Carolina State University. [date unknown]. Sweetpotato varieties. Sweetpotato and Potato Breeding and Genetics Programs. https://potatoes.cals.ncsu.edu/sweetpotato/sweetpotato-varieties/.
Parada C, Quesada-Ocampo L. 2023. How to identify and manage sweetpotato scurf caused by Monilochaetes infuscans. North Carolina State University Extension. https://plantpathology.ces.ncsu.edu/2021/03/how-to-identify-and-manage-sweeetpotato-scurf-caused-by-monilochaetes-infuscans/.
Pehling D. 2013. Pests: Vole management in home backyard gardens (FS094E). Washington State University Extension. https://s3.wp.wsu.edu/uploads/sites/2071/2014/04/Vole-Mgmt-Backyard-Gardens-FS094E.pdf.
Poggi S, Le Cointe R, Lehmhus J, Plantegenest M, Furlan L. 2021. Alternative strategies for controlling wireworms in field crops: A review. Agriculture-Basel. 11(5):436. https://doi.org/10.3390/agriculture11050436.
Pscheidt J, Putnam M. 2023. Fasciation. Pacific Northwest Pest Management Handbooks. https://pnwhandbooks.org/plantdisease/pathogen-articles/common/fasciation.
Quinn N. 2024. Pest notes: Rats (UC ANR Publication 74106). University of California Cooperative Extension. https://ipm.ucanr.edu/home-and-landscape/rats/pest-notes/#gsc.tab=0.

The information in this publication is based upon work that is supported by the U.S. Department of Agriculture, National Institute of Food and Agriculture, under award number 2022-38640-37490 through the Western Sustainable Agriculture Research and Education Program under project number WPDP25-003, and Hatch Project WNP0010 Accession 7005372. Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the author(s) and are not meant to endorse any businesses or detract from any not listed.